  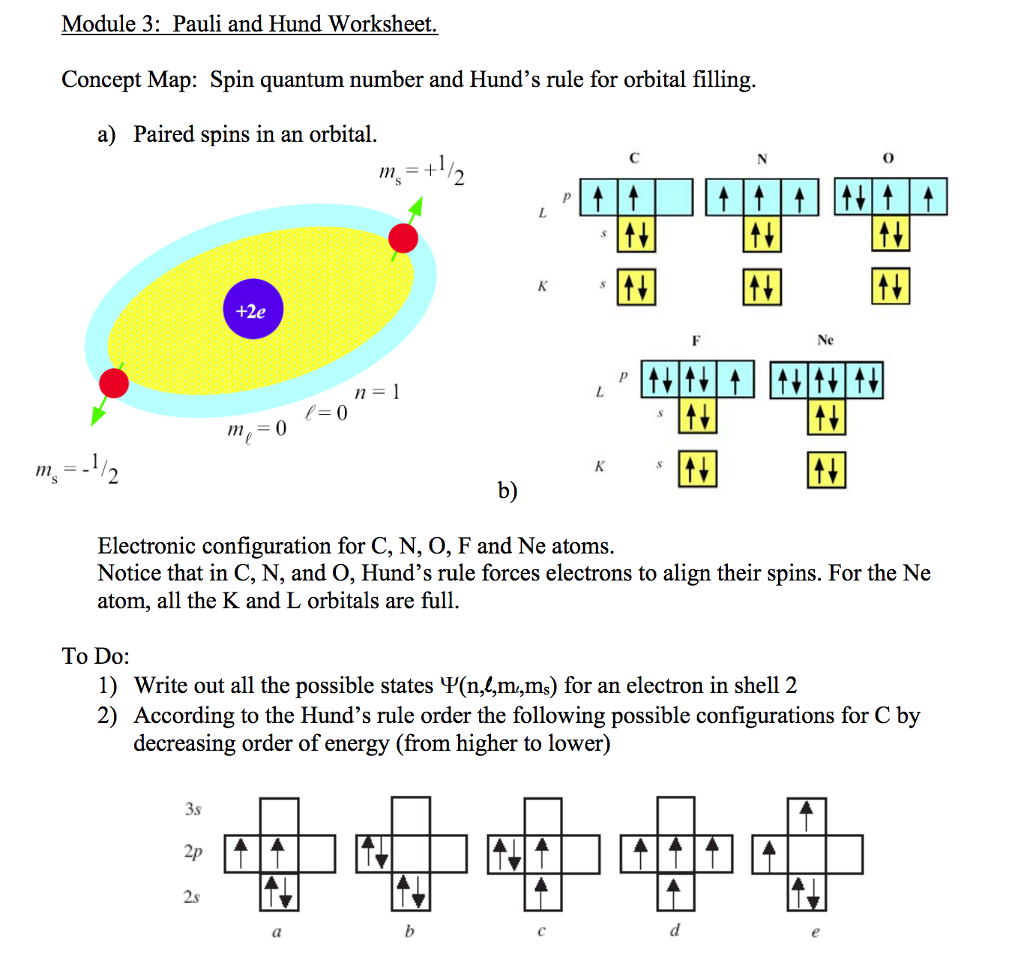
For instance, you could never have the following possible quantum numbers for an atom: The quantum numbers also come with a set of restrictions which inherently gives you useful information about where electrons will NOT be. Of course, with a little more effort, you could predict the exact location where the electron goes but in my example above, you know for a fact you could describe it using the quantum number formalism. You can do this by writing out all the possible permutations of the four quantum numbers. Where will the electron(s) go? Regardless of what wavelength of light we use, we know that we can describe the final location(s) using the four quantum numbers. Given a wavelength of light for example, one can excite carbon in any number of ways. Of course carbon is not always going to be in the ground state. The four quantum numbers allows us to communicate this information numerically without any need for a wordy description. We could communicate with each other that an electron is "located in the lowest energy, spherical atomic orbital" but it is much easier to say a spin-up electron in the 1$s$ orbital instead. The quantum numbers have been laid out in the same way. You could describe your home location any number of ways (GPS coordinates, qualitatively describing your surroundings, etc.) but we've adapted to a particular formalism in how we describe it (at least in the case of mailing addresses). We can write out the physical 'address' of these electrons in a ground-state configuration using quantum numbers as well as the location(s) of these electrons when in some non-ground (i.e. When an atom is in the ground state, these electrons will reside in the lowest energy orbitals possible (e.g. Electrons reside in atomic orbitals (which are very well defined 'locations'). Therefore we could say that you can be found in either of these locations depending on the time of day. You may begin your day at your home address but if you have an office job, you can be found at a different address during the work week. We can narrow the scope of this analogy even further. 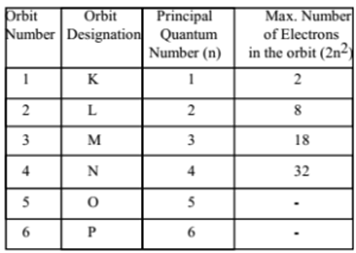
It allows one to pinpoint your exact location out of a set of $n$ locations you could possibly be in. Think about it as the mailing address to your house. Quantum numbers full#A full set of quantum numbers describes a unique electron for a particular atom. Quantum numbers give information about the location of an electron or set of electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
